|
We recently identified two novel protein O-glucosyltransferases, Protein O-glucosyltransferase 2 (POGLUT2) and 3 (POGLUT3), that modify a small fraction of EGF repeats on Notch. Mutations within the 47 epidermal growth factor-like (EGF) repeats of FBN1 cause autosomal dominant disorders including Marfan Syndrome which is characterized by disrupted TGF-β signaling. Through protein-protein interactions with latent transforming growth factor (TGF) β binding protein 1 (LTBP1), microfibrils regulate TGF-β signaling. 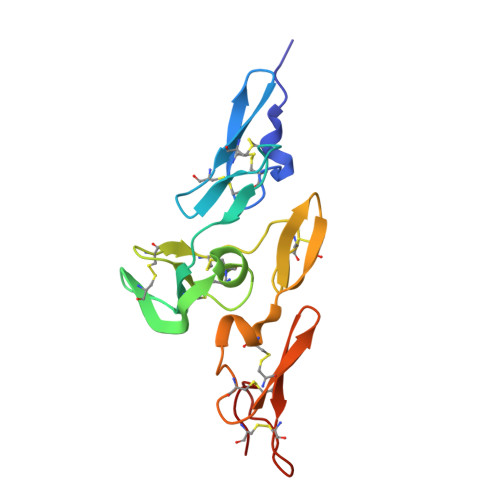
Other mutations cause the substitution of single amino acids in the calcium binding consensus sequence, which could affect calcium binding but also the hydroxylation of Asp/Asn residues or the modification of Ser residues.įibrillin-1 (FBN1) is the major component of extracellular matrix microfibrils which are required for proper development of elastic tissues, including the heart and lungs. Most of these mutations have been found in pEGF domains, frequently substituting Cys for another amino acid, destroying the pEGF motif secondary structure along with its calcium binding potential. Marfan syndrome, a heritable disorder of connective tissue, is known to be associated with mutations in the FBN1 gene. The calcium binding consensus sequence also contains a conserved Ser residue with an apparently novel modification, which causes the Ser residue to behave like an Asp residue during protein sequencing. This is the first demonstration of this modification in a connective tissue protein. Five partially hydroxylated Asn residues have been identified by protein sequence analysis of fibrillin-1 fragments. 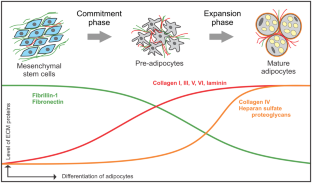
There is a second overlapping consensus sequence in the pEGF domains for beta-hydroxylation of a specific Asp/Asn residue. Scatchard plot analysis of the calcium binding data shows that PF2 has six to seven high affinity binding sites with a Kd = 250 microM at pH 7.5. Peptide sequence data and pepsin fragment size determination indicate that PF2 contains seven pEGF domains, each with the calcium binding consensus sequence. The calcium binding potential of a fibrillin-1 pepsin fragment (PF2) was quantitatively analyzed using microvolume equilibrium dialysis. 
Fibrillin-1 contains 43 precursor epidermal growth factor-like (pEGF) domains that have a consensus sequence for calcium binding. The extracellular matrix protein fibrillin-1 is a major component of elastic microfibrils, which are complex assemblies of several proteins and are found in most connective tissues, frequently associated with elastin.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
